
The granting of aid for the implementation of an investment plan to the investment body “THERACELL LABORATORIES IKE” was approved, with a total supported cost of 82,916,661 euros. The investment plan has been classified as a strategic investment.
According to the decision signed by Deputy Minister of Development and Investments Nikos Papathanasis, the investment concerns the clinical development and application of advanced autologous (the patient is the cell donor) cell and gene therapies, the production of which will be carried out near the treatment centers, within specially configured autonomous production OMPUL (Orgenesis Mobile Processing Unit Laboratories) units with the application of the decentralized POCare (Point of Care) model, i.e. the development, production and administration of CGTs (Cell & Gene Therapies) close to the patient.
The object
As noted, the object of the investment is an initial investment in a new economic activity and includes:
a. the installation of OMPULs within the surrounding area, near the point of patient hospitalization, of selected hospitals that have the required space,
b. the completion of industrial research and automation of OMPULs for mass production of treatments,
c. the experimental development of treatments through clinical trials within the framework of the approval process which will concern the testing and documentation of new products in an environment of real conditions of diseases and hospitalization, and
d. the production and distribution of new cell and gene therapies.
Plan
The investment plan of “THERACELL LABORATORIES IKE” was characterized, by decision of the Interministerial Committee for Strategic Investments was included in the “Strategic Investments 2” category, i.e. strategic investments which create in a sustainable way at least 50 new Annual Work Units (AWU) and their total budget is greater than 30,000,000 euros, according to the relevant legal provisions. That is, the approved aid falls under the category of regional investment aid and the category of aid for research and development projects.
Read also: Strategic Investments: The Selection Process, Duties and Fees of Auditors
The beneficiary of the aid is the company under the name “THERACELL LABORATORIES IKE” in which “THERACELL ADVANCED BIOTECHNOLOGICAL APPLICATIONS SOLE REPRESENTATIVE COMPANY” and ORGENESIS MARYLAND INC each participate by 50%.
The corporate capital of the investment entity at the time of submission of the affiliation application amounts to 25,000 euros.
Cost of investment plan
The subsidized expenses that were accepted upon the recommendation of “Enterprise Greece”, based on the submitted investment file, are divided into investment expenses, amounting to 4,349,603 euros, and expenses for research and development, amounting to 78,567,058 euros. The total cost of the investment project amounts to 82,916,661 euros.
Aid provided
– Tax Incentives
The incentive of tax exemption for the enhanced costs of the laboratory in Corinth and the 7 OMPULs of the investment plan, amounting to 4,349,603.00 euros, is debited. The amount of the granted tax exemption aid amounts to 1,957,321 euros, i.e. 45% of the total expenditure for which aid is granted.
– Aid for research and development
The incentive of the grant for research and development projects for the enhanced costs of experimental development, industrial research and feasibility studies of the investment plan, amounting to 78,567,058 euros, is debited. The amount of the given grant for research and development costs amounts to 31,728,747 euros.
Funding sources
The financial structure of the investment plan is analyzed by funding source as follows:
A. Equity
– Increase in corporate capital of 4,145,833 euros
B. External financing
– Bond loan of 37,000,000 euros
– Bank loan of 10,042,081 euros
Total Own Participation 51,187,914 euros
The investment body’s own participation, whether through equity or external financing, must not contain any state aid, public support, or provision.
The amount of funds per funding source will be determined precisely upon completion of the investment plan and will be calculated at this actual cost.
Conditions for granting said aid
The support of the investment plan is approved on the condition that the investment entity complies with the following conditions.
1. Place of installation
The investment will be implemented in a leased industrial property at the 80th km of Athens – Corinth highway, in the Municipality of Corinth, where a research and development laboratory for future treatments will be built.
The autonomous production units (OMPULs) will be temporarily placed in selected areas within or near hospitals and in any case closer to the point of patient hospitalization. The investment body is obliged, during the implementation control of the investment, to provide documents for the use of the plots on which the above autonomous production units will be installed.
2. Dependent job positions
The investment body is a newly established company and at the time of submission of the application for inclusion in the procedures of Law 4608/2019 did not employ any staff.
The new positions that will be created from the first year of full operation of the investment amount to 174 annual work units.
The application submission date is May 7, 2021, which is also the date on which the start of work on the investment plan can take place, and the completion of the investment plan is set for December 31, 2026.
Among the obligations of the investment implementing body is that it must comply with the long-term obligations after the completion of the investment plan and the certification of the start of production for a period of 3 years.
Latest News

Airbnb: Greece’s Short-Term Rentals Dip in March Amid Easter Shift
Data from analytics firm AirDNA shows that average occupancy for short-term rentals dropped to 45% in March, down from 49% the same month last year.

Easter Week in Greece: Holy Friday in Orthodoxy Today
At the Vespers service on Friday evening the image of Christ is removed from the Cross and wrapped in a white cloth

Meloni and Trump Meet in Washington, Vow to Strengthen Western Ties
“I am 100% sure there will be no problems reaching a deal on tariffs with the EU—none whatsoever,” Trump stressed.

ECB Cuts Interest Rates by 25 Basis Points in Expected Move
The ECB’s Governing Council opted to lower the deposit facility rate—the benchmark for signaling monetary policy direction—citing an updated assessment of inflation prospects, the dynamics of underlying inflation, and the strength of monetary policy transmission.

Current Account Deficit Fell by €573.2ml Feb. 2025: BoG
The improvement of Greece’s current account was mainly attributed to a more robust balance of goods and, to a lesser extent, an improved primary income account

Hellenic Food Authority Issues Food Safety Tips for Easter
Food safety tips on how to make sure your lamb has been properly inspected and your eggs stay fresh.

Greek Kiwifruit Exports Smash 200,000-Ton Mark, Setting New Record
According to data by the Association of Greek Fruit, Vegetable and Juice Exporters, Incofruit Hellas, between September 1, 2024, and April 17, 2025, kiwifruit exports increased by 14.2%.

Easter Tourism Boom: Greece Sees 18.3% Surge in Hotel Bookings
Among foreign markets, Israel has emerged as the biggest growth driver, with hotel bookings more than doubling—up 178.5% year-on-year.
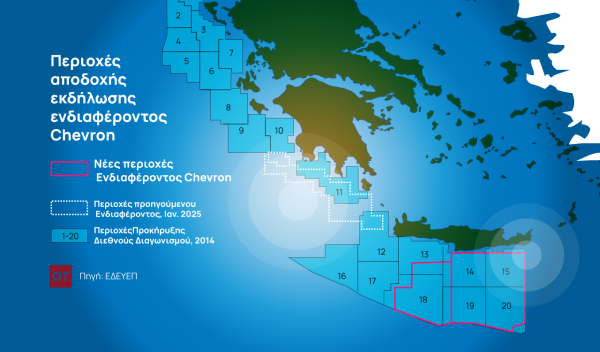
Greece to Launch Fast-Track Tender for Offshore Hydrocarbon Exploration
Last week, Papastavrou signed the acceptance of interest for the two Cretan blocks, while similar decisions regarding the two Ionian Sea blocks were signed by his predecessor

American-Hellenic Chamber of Commerce to Open Washington D.C. Branch
AmCham's new office aims aims to deepen U.S.-Greece economic ties and promote investment and innovation between the two countries







![Πλημμύρες: Σημειώθηκαν σε επίπεδα ρεκόρ στην Ευρώπη το 2024 [γράφημα]](https://www.ot.gr/wp-content/uploads/2025/04/FLOOD_HUNGRY-90x90.jpg)




![Airbnb: Πτωτικά κινήθηκε η ζήτηση τον Μάρτιο – Τι δείχνουν τα στοιχεία [γράφημα]](https://www.ot.gr/wp-content/uploads/2024/07/airbnb-gba8e58468_1280-1-90x90.jpg)

























![Airbnb: Πτωτικά κινήθηκε η ζήτηση τον Μάρτιο – Τι δείχνουν τα στοιχεία [γράφημα]](https://www.ot.gr/wp-content/uploads/2024/07/airbnb-gba8e58468_1280-1-600x500.jpg)


 Αριθμός Πιστοποίησης
Αριθμός Πιστοποίησης